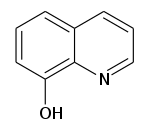
What Is Hydroxyquinoline Used For?
 | |
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name Quinolin-eight-ol | |
| Other names 1-Azanaphthalene-8-ol, Fennosan H xxx, Hydroxybenzopyridine, Oxybenzopyridine, Oxychinolin, Oxyquinoline, Phenopyridine, Quinophenol, Oxine, 8-Quinolinol | |
| Identifiers | |
| CAS Number |
|
| 3D model (JSmol) |
|
| ChEBI |
|
| ChEMBL |
|
| ChemSpider |
|
| ECHA InfoCard | 100.005.193 |
| KEGG |
|
| PubChem CID |
|
| UNII |
|
| CompTox Dashboard (EPA) |
|
| InChI
| |
| SMILES
| |
| Properties | |
| Chemical formula | C9H7NO |
| Molar mass | 145.16 g/mol |
| Advent | White crystalline needles |
| Density | ane.034 k/cm3 |
| Melting point | 76 °C (169 °F; 349 Thousand) |
| Boiling point | 276 °C (529 °F; 549 One thousand) |
| Pharmacology | |
| ATC code | G01AC30 (WHO) A01AB07 (WHO) D08AH03 (WHO) R02AA14 (WHO) |
| Hazards | |
| Occupational safe and wellness (OHS/OSH): | |
| Primary hazards | flammable |
| Condom data sheet (SDS) | External MSDS |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
8-Hydroxyquinoline (as well known equally oxine) is a chelating agent which has been used for the quantitative determination of metal ions.
In aqueous solution eight-hydroxyquinoline has a pKa value of ca. ix.ix[i] It reacts with metallic ions, losing the proton and forming 8-hydroxyquinolinato-chelate complexes.
The aluminium complex[2] is a common component of organic light-emitting diodes (OLEDs). Variations in the substituents on the quinoline rings affect its luminescence properties.[3]
In the photochemically induced excited-land zwitterionic isomers are formed in which the hydrogen atom is transferred from oxygen to nitrogen.[4]
The complexes as well as the heterocycle itself exhibit antiseptic, disinfectant, and pesticide properties,[five] [6] functioning every bit a transcription inhibitor.[vii] Its solution in booze is used in liquid bandages. Information technology one time was of interest equally an anti-cancer drug.[viii]
A thiol analogue, 8-mercaptoquinoline is as well known.[9]
The roots of the invasive institute Centaurea diffusa release 8-hydroxyquinoline, which has a negative effect on plants that have not co-evolved with information technology.[10]

Run into too [edit]
- Clioquinol, an antifungal drug and antiprotozoal drug.
- PBT2
- QUPIC
- Ionophore
- Trace metal detection test
References [edit]
- ^ Albert, A.; Phillips, J. Due north. (1956). "264. Ionization Constants of Heterocyclic Substances. Part Two. Hydroxy-Derivatives of Nitrogenous Vi-Membered Ring-Compounds". Journal of the Chemical Society (Resumed). 1956: 1294–1304. doi:10.1039/JR9560001294.
- ^ Katakura, R.; Koide, Y. (2006). "Configuration-Specific Synthesis of the Facial and Meridional Isomers of Tris(viii-hydroxyquinolinate)aluminum (Alq3)". Inorganic Chemical science. 45 (15): 5730–5732. doi:10.1021/ic060594s. PMID 16841973.
- ^ Montes, V. A.; Pohl, R.; Shinar, J.; Anzenbacher, P. Jr. (2006). "Effective Manipulation of the Electronic Effects and Its Influence on the Emission of 5-Substituted Tris(8-quinolinolate) Aluminum(III) Complexes". Chemistry: A European Journal. 12 (17): 4523–4535. doi:ten.1002/chem.200501403. PMID 16619313.
- ^ Bardez, E.; Devol, I.; Larrey, B.; Valeur, B. (1997). "Excited-Land Processes in 8-Hydroxyquinoline: Photoinduced Tautomerization and Solvation Effects". The Journal of Concrete Chemistry B. 101 (39): 7786–7793. doi:10.1021/jp971293u.
- ^ Phillips, J. P. (1956). "The Reactions of 8-Quinolinol". Chemical Reviews. 56 (2): 271–297. doi:10.1021/cr50008a003.
- ^ "viii-Hydroxyquinoline". Medical Dictionary Online. Archived from the original on 2016-x-09. Retrieved 2016-03-09 .
- ^ "8-Hydroxyquinoline". Sigma-Aldrich. Retrieved 2022-02-fifteen .
- ^ Shen, A. Y.; Wu, S. N.; Chiu, C. T. (1999). "Synthesis and Cytotoxicity Evaluation of some viii-Hydroxyquinoline Derivatives". Journal of Chemist's and Pharmacology. 51 (v): 543–548. doi:10.1211/0022357991772826. PMID 10411213. S2CID 33085238.
- ^ Fleischer, Holger "Structural chemical science of complexes of (n - 1)d10nsk metal ions with β-N-donor substituted thiolate ligands (m=0, 2)" Coordination Chemistry Reviews 2005, book 249, pp. 799-827. doi:10.1016/j.ccr.2004.08.024
- ^ Vivanco, J.K.; Bais, H.P.; Stermitz, F.R.; Thelen, G.C.; Callaway, R.M. (2004). "Biogeographical variation in customs response to root allelochemistry: novel weapons and exotic invasion". Environmental Letters. 7 (4): 285–292. doi:ten.1111/j.1461-0248.2004.00576.ten.
- ^ Cölle, M.; Dinnebier, R. E.; Brütting, W. (2002). "The structure of the blueish luminescent δ-stage of tris(eight-hydroxyquinoline)aluminium(3) (Alq3)". Chemical Communications. 2002 (23): 2908–9. doi:10.1039/b209164j. PMID 12478807.
What Is Hydroxyquinoline Used For?,
Source: https://en.wikipedia.org/wiki/8-Hydroxyquinoline
Posted by: covingtonalivink1991.blogspot.com


0 Response to "What Is Hydroxyquinoline Used For?"
Post a Comment